|
Indicates the date the device is no longer held or offered for sale by the labeler on record. Indicates the date the DI Record is published and available via Public Search. NATRELLE INSPIRA SCM 310CC MODERATE PROFILE |SMOOTH|ROUND|SILICONEĪdditional relevant information about the device that is not already captured as a distinct GUDID data attribute. The catalog, reference, or product number found on the device label or accompanying packaging to identify a particular product. The version or model identifies all devices that have specifications, performance, size, and composition within limits set by the labeler. The version or model found on the device label or accompanying packaging used to identify a category or design of a device. The brand name is the name that is typically registered with USPTO and have the ® and/or TM symbol. This information may 1) be on a label attached to a durable device, 2) be on a package of a disposable device, or 3) appear in labeling materials of an implantable device. The Proprietary/Trade/Brand name of the medical device as used in device labeling or in the catalog. NATRELLE INSPIRA COHESIVE BREAST IMPLANTS  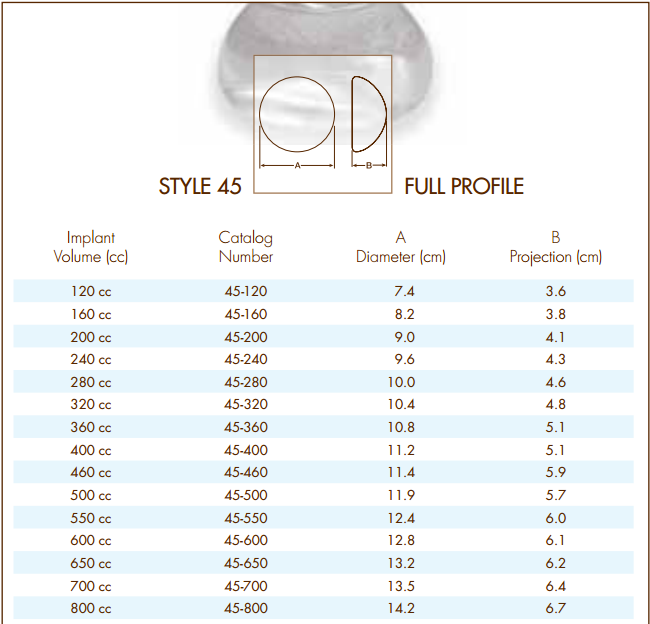 
Company name associated with the labeler DUNS Number entered in the DI Record.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
